by NOELLE NICOLLS
khalilanicolls@gmail.com
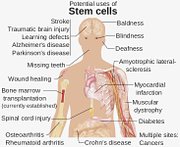
IMAGINE living in a world where a drug-free fix for HIV/AIDS existed, or a cure for cancer, blindness, Parkinson’s, diabetes, heart disease, even Lou Gehrig’s disease. Advocates of stem cell research are driven by this vision. They say the promise of stem cell therapy is the birth of a new medical paradigm more revolutionary than the advent of the internet.
Some even believe stem cell therapy is the key to immortality. As a result, those pursuing medical breakthroughs in the field are often driven by an age old obsession to find the magical elixir of life.
However, it is not just anti-aging obsessives who support stem cell research. Legitimate, professional medical researchers have sound reasons to be optimistic about the positive impact stem cell treatments could have on public health.
There are also investors who want to cash in on new medical breakthroughs. There are also patients suffering from incurable diseases or chronic disorders praying for stem cell therapy to be their miracle cure.
Debate on The Bahamas’ potential foray into the world of stem cell research and regenerative medicine has touched on some of the moral and ethical issues, but there has been virtually no debate of the economics of stem cell research. The government’s economic claims have gone unchallenged.
For the most part conversation has been hijacked by talk about a millionaire fashion designer who is very interested in the use of stem cells for anti-aging treatments and happens to be an investor in the Bahamas. When debate resumes in the House of Assembly next week, I hope it will turn to more substantive issues.
Despite all of the reasons that make stem cell research an important area of medical study, it remains controversial. ‘Frankenstein’ scenarios aside (as stem cells could unlock the key to human cloning) the primary reason for controversy is concern over the harvesting of human embryonic stem cells, which are highly favoured among all types of stem cells.
Embryonic stem cells are harvested from human embryos when they are only days old. These embryos are usually destroyed as the cells are extracted. Most religious institutions have strong moral objections to the harvesting of embryonic stem cells, arguing for the sanctity of life, even at the embryonic stage. However, many support the use of adult stem cells and amniotic stem cells, which come from umbilical cord tissue.
In countries that allow the harvesting of embryonic stem cells - Finland, France, Greece, the Netherlands, Sweden, Italy and the United Kingdom - these cells are obtained from surplus embryos at in vitro fertilisation (IVF) clinics.
Last year, the government’s task force took a stand against the use of controversial embryonic stem cells in the Bahamas, while sanctioning the use of adult and amniotic stem cells.
Although the committee first reported its findings late last year, debate reignited when the government tabled a Stem Cell Research and Therapy Bill along with draft regulations in the House of Assembly in April.
The new law will create a regulatory framework covering all aspects of stem cell research, including clinical and pre-clinical research, the use of non-human animal research subjects; and therapeutic uses in people.
The opposition’s main argument against supporting stem cell research in the Bahamas is concern over the proper enforcement of laws governing the industry. In our country of lax government regulation, the concern is valid; perhaps not sufficient to sabotage the government’s efforts, but valid nonetheless, given the high risks posed by the industry.
It is certainly plausible that one of the reasons the Bahamas is perceived as a favourable destination for stem cell research, when it does not have competitive infrastructure to supporting an industry of medical research, is its lax regulatory environment. It is certainly plausible that unscrupulous researchers, investors or medical practitioners could prey on the Bahamas while advancing their own personal agendas.
We all know a little too well that having a law on the books in the Bahamas is not the same as having a strong regulatory environment with robust systems of oversight. But the illusion of a legal framework could provide the prefect cover for unethical players.
The economics of medical research
If we put concerns about government oversight aside for the moment, and questions of morality and ethics; if we take the government at its word that its interest in stem cell research is not some kind of political payback, or at least a political nod, to Mr Nygard for alleged political contributions, there is still one glaring issue that has gone unchallenged.
It is the economics of medical research and the economics of stem cell medicine.
The Stem Cell Research Bill has been called “revolutionary”. It has been portrayed as having the power to “catapult the health sector” in the Bahamas and revolutionise medicine. When Prime Minister Perry Christie tabled the bill, he said stem cell projects “promise to attract leading scientists” who will relocate to the Bahamas and “conduct research, establish institutions and centres of excellence”. Mr Christie said many young Bahamian scientists and physicians will gain new skills and new opportunities. That the industry will generate new jobs at many levels, and will fuel accelerated growth in the economy. The industry will have spillover effects on all areas in the economy, including the tourism industry.
“Our young Bahamian scientists and doctors will be able to establish thriving professional lives right here in The Bahamas. They will then be able to contribute back to our community in the years to come, as well as to the international community,” said Mr Christie.
The government has boasted that stem cell research could inject more than $100 million into the Bahamian economy annually. Peter Nygard has touted the idea that a stem cell centre in the Bahamas could raise to the status of the Mayo Clinic in Rochester, which caters to thousands of people around the world and employs some 50,000 people.
In the context of a stagnant economy, these words sound like to me a Junkanoo orchestra: sweet to the ears. Rhetorically, it is easy to connect the dots between stem cell research, regenerative medicine, medical tourism and economic development for the Bahamian people. At the rhetorical level, the relationship between these four elements represents a magic formula for pandering to populist sentiment.
Who wouldn’t want to be associated with a medical breakthrough that could cure all of the world’s ills? Who doesn’t like the sound of an economic stimulus that requires no investment on the part of the government, only the simple passage of a new bill?
In reality, sweet words do not make for sound economic policy.
There are deep, uncharted chasms between each step on the trajectory from private investment in stem cell research to economic profit for the Bahamas as a whole.
I am bewildered by the level of hype being generated around the issue, when the evidence does not seem to support many of claims being made, and our national needs do not justify them.
Let us unpack the economic issues first.
In the United States, studies show medical research has a positive economic impact. However, much of that success is attributed to a private/public sector funding partnership. Government funding accounts for approximately 36% of total research funding, according to some estimates. In 2003, for example, the National Institute of Health (NIH), the largest federal agency of medical research, was responsible for 28% (about US$26.4 billion) of the total biomedical research funding spent annually in the US. In 2009, the NIH injected $15.6 billion research dollars into medical schools and teaching hospitals alone. In places where medical research is highly successful from an economic impact point of view, there is a large sum of government money being spent. Unless I missed it, I have not heard the Bahamian government talking about investing in stem cell research or regenerative medicine, only legislating it.
The Association of American Medical Colleges (AAMC) published a 2009 report, summarising the economic and employment impact from federal and state funded research in the 25 states (plus the District of Colombia). Number one on the list: California (population 38 million). The total direct and indirect economic impact of the medical research industry for California from the AAMC network was $5,360 million (as in $5.3 billion). The employment impact on the other hand was only 35,734. Number 25 on the list was Iowa. With a population of 3 million people, the economic impact on Iowa was $400 million; while the employment impact was 2,719 jobs.
If our government is projecting a $100 million industry for the Bahamas (with a population of 0.4 million), where does that really place us in the grand scheme of things? $100 million sounds like a large number at first glance, but further analysis is required to assess its true value. Does $100 million reflect a projection of total spending, total revenue, total tax revenue, all of the above? And how many permanent jobs would an industry of this size actually produce? Is the $100 million projection speaking specifically to stem cell research or also stem cell therapy, because research and clinical treatment are two different things. Stem cell research is still in its infancy, which means stem cell treatment is still in a state of trial; it is not practiced widely at the clinical level. So I ask again, what exactly is the economic model?
In California, $1.5 billion in committed research grants to the California Institute for Regenerative Medicine is generating $286 million in new tax revenue for the state and local governments through 2014, according to a study conducted by Emeryville-based global consulting firm Berkley Research Group. CIRM was established by the state government in 2004 with the passage of the California Stem Cell Research and Cures Act.
It released an independently produced economic impact study measuring the stem cell agency’s financial impact on the state. The study found that the same grants will generate an estimated $530 million in tax revenue for the federal government.
There are models of success out there, but it goes without saying, California’s success, for example, is not a blueprint for the Bahamas. The markets are completely different. For one, California has the infrastructure to support a medical research industry: legal framework, sources of funding, technical expertise, supply chain, demand for services. Without the infrastructure or “risk-sharing partnerships”, any high risk industry will likely operate at 100 times the cost with 100 times the liability.
In the Bahamas, a few private ventures that represent isolated pockets in a global industry (no matter how world class they may be) will not automatically propel the Bahamas into global dominance. We should have learned that lesson from the tourism industry.
Bahamas Heart Centre
As a case study, let us look at the Bahamas Heart Centre (BHC), which entered the record books last year by performing the Bahamas’ first cardiac stem cell implantation in conjunction with Advanced Innovative Medicine Inc of Orlando, Florida. A very advanced team of doctors treated a 62-year-old cardiac patient who had advanced coronary artery disease by implanting some of the patient’s own stem cells into his heart. The patient, a ‘medical tourist’, had already undergone by-pass surgery on a number of occasions, after his first heart attack in 1989. The stem cell treatment was considered a “last option” before considering a heart transplant. He was eligible for this experimental treatment after meeting “a very strict set of criteria” to qualify, based on rigid clinical research trial protocols.
I congratulate the Bahamas Heart Centre on this successful treatment, and should they be able to save more lives with their stem cell therapy I support them doing so. However, I would be interested in speaking with the BHC’s chief financial officer. I highly doubt the medical facility has plans to change its business model to invest everything it has in stem cell treatment.
For not-so-sinister business reasons, offshore clinical trials are being promoted in places like China by leading centres of stem cell research for the same reasons corporate America outsources many of its business processes. In fact, China has picked up traction in the stem cell research race. So perhaps the Bahamas could become a hub for offshore clinical trials if it could help businesses lower their development costs. In this light, the BHC’s work can also be seen as a success.
Nonetheless, stem cell treatment for the foreseeable future is likely to be a small line item in the centre’s over revenue model, I would suggest.
Big Pharma
It might be the star player in the BHC’s research and development or clinical trials department, but it is not likely to be the foundation of their business model. Simply put, “the stem cell and regenerative medicine market is still in its commercial infancy” despite the scope of opportunities that exist. This, according to the Stem Cell Network (SCN), a Canadian not-for-profit corporation that funds applied stem cell research and facilitates collaboration between universities, industry, government and non-governmental organisation.
According to the SCN, Big Pharma is not even on board with stem cell medicine, because the economics are too uncertain.
“Cell-based biologic products represent both new technology and a business model that remains largely unknown, but is certainly different from traditional drug or device development. Drug and device companies are proceeding cautiously, and waiting to see who emerges from the 100 or more stem cell companies now operating around the world, most of which tend to lack the critical mass and the clinical, regulatory and manufacturing capabilities to establish a sustainable product portfolio and technology pipeline,” states SCN.
Former Fortune senior editor Jeffrey M. O’Brien publish a 2012 article investigating the stem cell business. His research showed that “shares in almost any public stem cell company can be had for less than $1. One of the highest fliers, Stem Cells Inc. (STEM), reached $171 a share once upon a time. In mid-September it was trading at 95�.”
The first-ever clinical trial for embryonic-stem-cell therapy approved by the Federal Drug Administration (FDA) in the United States was conducted by Geron, a leading Silicon Valley biotech company. Geron injected four spinal-cord patients with its stem-cell therapy. The treatment had previous success enabling paraplegic rats to walk. The company shut down its clinical stem cell trial months after it began, blaming difficult economic conditions. Reports indicated “the treatment did not show the immediate promise many had hoped”. The company, however, said it wanted to concentrate resources on two new and promising cancer drugs”.
Advanced Cell Technology (ACTC), another biotech company, picked up the slack. Its chief scientist Robert Lanza has made substantial progress in treating a type of blindness (macular degeneration), a condition that affects tens of millions of people, where spinal-cord injuries only represent a market of tens of thousands. ACTC has not figured out the economic model either. One report said ACT “has been scratching and clawing to survive for years”, dealing with a range of problems from raising venture funding to settling patent-infringement lawsuits.
When ACTC received FDA approval to commence clinical trials in 2010, it had already “spent upwards of $100 million on research”, according to reports.
No matter which way you spin it, the economics do not seem to be there - at the moment. For the Bahamas, stem cell medicine could be a hyped-up experiment with an empty payoff or it could be a forward thinking move by a bold government. Either way, it is a matter that could have been handled with much less fanfare and controversy.
Dangers of distraction
Debate about the stem cell bill has become a colossal national distraction, not because discussing stem cell medicine itself is a waste of time; but its advocates seem incapable of presenting sober arguments that tell the full picture; and they seem motivated by the hype. Debate has degraded into political squabbling, dreams of immortality and ‘Frankenstein’ science.
There is no doubt that advanced breakthroughs in clinical stem cell therapy could transform the Bahamas and the world. There is hardly anything to debate when it comes to the medical possibilities; they are bewildering and awe-inspiring.
But what is most relevant to the debate at this time is not the dream of a brave new world where incurable diseases are as obsolete as floppy disks, it is the notion that stem cell research and regenerative medicine can provide a viable economic stimulus for the Bahamas. It would be good if our leaders could sit across the aisle like adults and debate serious issues with competence, restraint and intellectual honesty.
As if the economics of stem cell medicine were not enough to fully explore the matter, there is also the matter of establishing our national health priorities. Next time, I plan to look at how our vigorous push for stem cell medicine contends with our national health priorities and the treatment opportunities that will open up for Bahamians in the near and long-term future.
I will also look at the government’s obsession with “revolutionary” projects that never live up to their promise. Remember the former government’s telemedicine project, which was also supposed to be a revolutionary technology that would transform health care in the Bahamas and put us on the map?
I support stem cell research in theory, and if we are to venture into the area (as private companies have already done in the Bahamas), there should be government regulation. But I do not support stem cell research becoming a national distraction. When debate resumes in the House of Assembly next week, I caution the government, as the old people say, when you lie with dogs you catch fleas. My advice to the government is to approach the conclusion of this stem cell debate with sobriety and restraint. Not everyone in the Bahamas deals in dreams. Some of us deal in fact.
Noelle Nicolls is the Tribune’s Features Editor. She is also a travel writer, women’s activist and entrepreneur. Follow her on Twitter @noelle_elleon. For questions or comments, email khalilanicolls@gmail.com




Comments
Marthasmith 8 years, 11 months ago
Using stem sells in medical purpose already shows great promise, it would be a shame not to keep developing the technology. With a bit more research and data process help from http://microlabinfo.com/equipment/spe...">http://microlabinfo.com/equipment/spe... , the scientists will be able to come up with more and more developed treatments for all kind of diseases.
isabella 8 years, 10 months ago
Stem cell therapy would definitely bring many positive changes in the field of healthcare. However, there should be more research work on this which could help people find the right cure. Now many advanced medication and medical devices are available online. Not just that but one can also find medical scrubs for healthcare workers online at http://www.uniformstoyou.com/healthca...">http://www.uniformstoyou.com/healthca....
Sign in to comment
Or login with:
OpenID